G protein linked receptors1/31/2024 The selectivity of β-adrenoceptor antagonists at the human β1, β2 and β3 adrenoceptors. Allosteric modulators of GPCRs: a novel approach for the treatment of CNS disorders. GPCRDB information system for G protein-coupled receptors. Modulation of the proline kink in transmembrane 6 by a rotamer toggle switch. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein coupled receptors. Structure of bovine rhodopsin in a trigonal crystal form. The retinal conformation and its environment in rhodopsin in light of a new 2.2 Å crystal structure. This paper presents the first three-dimensional crystal structure of a G-protein-coupled receptor, the visual photoreceptor rhodopsin. Crystal structure of rhodopsin: A G-protein-coupled receptor. 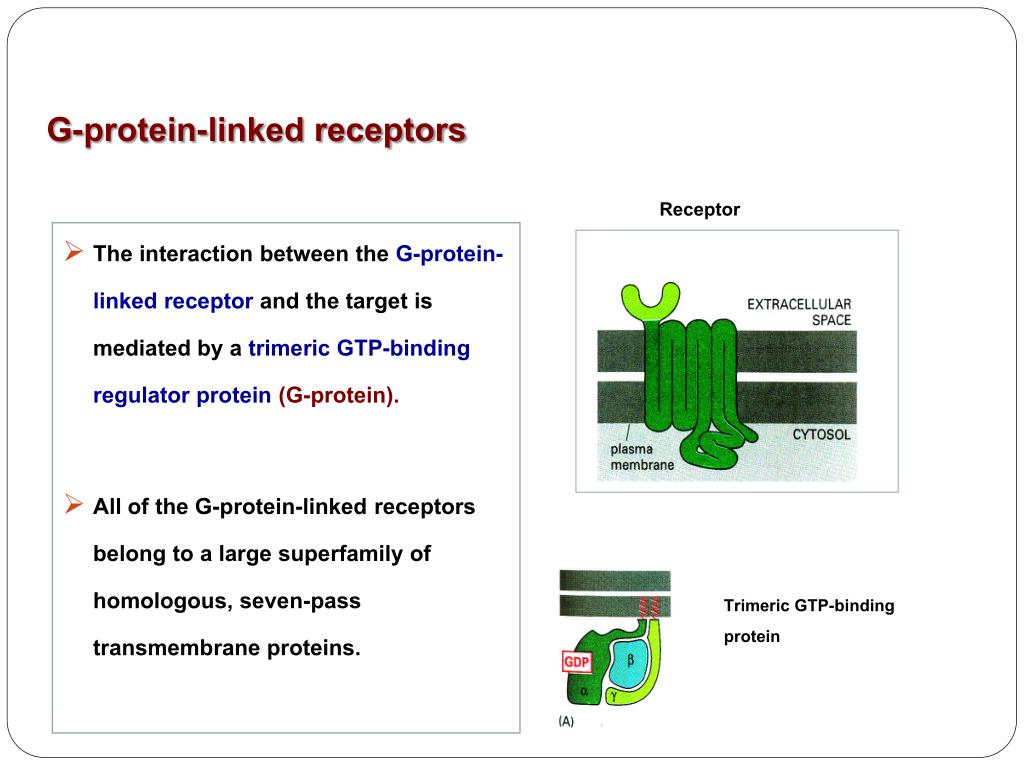
The 2.6 angstrom crystal structure of a human A2A adenosine receptor bound to an antagonist. Structure of a β1-adrenergic G-protein-coupled receptor. A specific cholesterol binding site is established by the 2.8 Å structure of the human β2-adrenergic receptor. 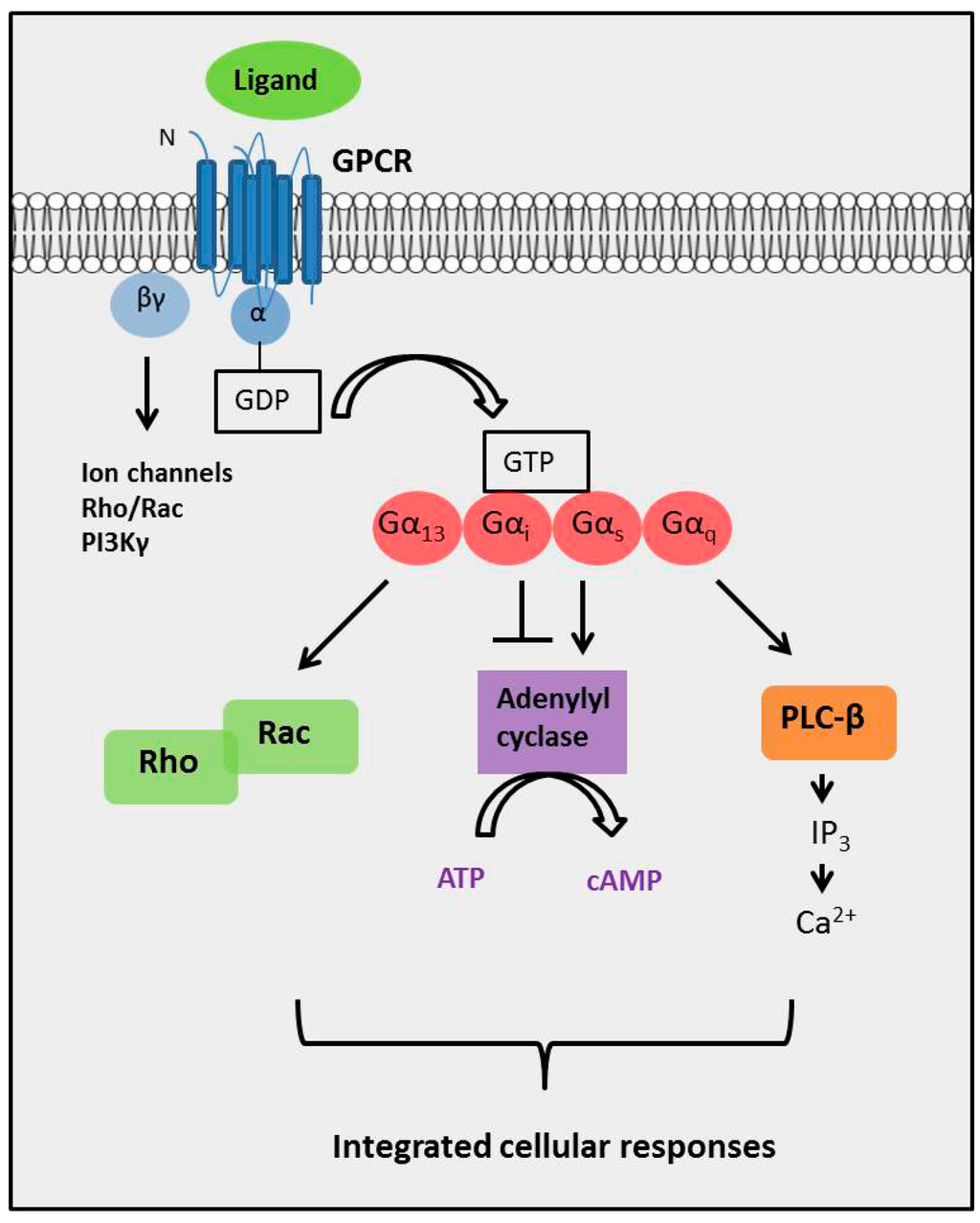
High-resolution crystal structure of an engineered human β2-adrenergic G-protein-coupled receptor. GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function. This is the first reported three-dimensional crystal structure of a ligand-activated G-protein-coupled receptor. Crystal structure of the human β2 adrenergic G-protein-coupled receptor. This paper presents the first three-dimensional structure of a G-protein-coupled receptor using cryoelectron microscopy of two-dimensional crystals. Characterisation of an improved two-dimensional p22121 crystal from bovine rhodopsin. Conformational complexity of G-protein-coupled receptors. A unique mechanism of beta-blocker action: carvedilol stimulates beta-arrestin signaling. The evasive nature of drug efficacy: implications for drug discovery. Probing the beta2 adrenoceptor binding site with catechol reveals differences in binding and activation by agonists and partial agonists. Kinetic evidence for intermediate conformational states. Sequential binding of agonists to the beta2 adrenoceptor. Agonist-induced conformational changes in the G-protein-coupling domain of the beta 2 adrenergic receptor. Caveolae and lipid rafts: G protein-coupled receptor signaling microdomains in cardiac myocytes. Roles of G-protein-coupled receptor dimerization. Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Desensitization of G protein-coupled receptors. Beta-arrestin-mediated activation of MAPK by inverse agonists reveals distinct active conformations for G protein-coupled receptors. Beta-arrestin-dependent, G protein-independent ERK1/2 activation by the beta2 adrenergic receptor. 
Recent advances in cardiac beta(2)-adrenergic signal transduction. Inverse agonist activity of beta-adrenergic antagonists. Negative antagonists promote an inactive conformation of the beta 2-adrenergic receptor. Samama, P., Pei, G., Costa, T., Cotecchia, S. This paper provides a comprehensive analysis of sequence relationships between G-protein-coupled receptors in the human genome. Phylogenetic analysis, paralogon groups, and fingerprints. The G-protein-coupled receptors in the human genome form five main families.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
